Therapeutic Function: CNS Stimulant AMINEPTINE HYDROCHLORIDE
Chemical Name: 7-[~10,11-Dihydro-5Hdibenzo[a~lcyclohepten-5-yl-amino] heptanoic acid hydrochloride
Common Name: -
Structural Formula:
(base)
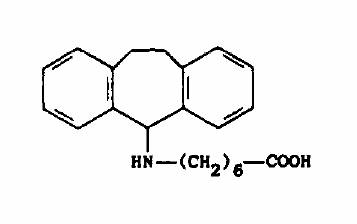
Chemical Abstracts Registry No.: 5757469-1 (Base); 30272483 (Hydrochloride)
Trade Name : Manufacturer : Country : Year IntroducedRaw Materials
Survector : Eutherapie : France : 1978
Survector : Servier : Italy : 1982
Maneon : Poli : Italy : 1982
5-Chloro-10,I 1 dihydrodHdibenzo(a,d)cycloheptene
Ethyl 7aminoheptanoateManufacturing Process
6.5 g of 5chloro-10,l ldihydrobHdibenzo(a,d)cycloheptene in 60 ml of nitromethane and 10.8 g of ethyl 7aminoheptanoate in 12 ml of nitromethane were mixed at ambient temperature. The reaction was slightly exothermic. The reaction mixture was left to stand overnight and the solvent was evaporated in vacuo. The residue was taken up in normal hydrochloric acid and the resulting precipitate was filtered off.10.5 g of crude ethyl 7-[dibenzo(a,d)cycloheptadieneb-yl] aminoheptanoate hydrochloride were obtained, of which a sample recrystallized from benzene gave a pure product melting instantaneously a t 166Oto 168OC.
The hydrochloride of the crude ester obtained above was added to 25 ml of 2 N hydrochloric acid. The whole was kept under reflux for 2 hours. The material dissolved and a new hydrochloride then reprecipitated. After cooling, the hydrochloride of the crude acid was filtered off, washed with iced water and then recrystallized from distilled water. 5.7 g of 7-[dibenzo(a,d)cycloheptadienb-yll aminoheptanoic acid hydrochloride were obtained, melting instantaneously at 226Oto 230'C.
References
Merck Index 409
Kleeman & Engel p. 40
DOT 19 ( I O ) 547 (1983)
I.N. p. 69
Melen ,C., Danree, B. and Poignant, J.C.; US. Patent 3,758,528; September 11,1973; assigned
to Societe en nom Collectif Science Union e t Cie; Societe Francaise de Recherche Medicale
Melen, C., Danree, B. and Poignant, J.C.; U.S. Patent 3,821,249; June 28, 1974; assigned to Societe en nom Collectif Science Union e t Cie; Societe Francaise de Recherche Medicale
HOMEAmineptine : refs
Amineptine: sources
Search amineptine.com
Federal Register Feb 2003
Product information (in French)